Electrochemical synthesis of polyaniline cross-linked NiMoO4 nanofibre dendrites for energy storage devices†
Abstract
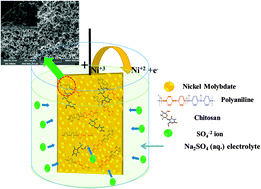
Electrochemical synthesis of polyaniline deposited on a graphite electrode using p-toluenesulfonic acid has been carried out via a potentiodynamic method. The feasibility to store energy in polyaniline has been demonstrated in 1 M Na2SO4 electrolyte using cyclic voltammetry and charge–discharge analyses. The obtained low capacitance (247 mF cm−2) for polyaniline limits its practical application. Therefore, as an alternative approach, aniline is polymerized in the presence of a novel non-ionic surfactant (Quillaja Saponin) and a marginal increase in capacitance to 342 mF cm−2 is observed. Adsorption of a surfactant on aniline resulted in a profound effect on its electrochemical behaviour. To further enhance the capacitance, innovatively, polyaniline is cross-linked with nickel molybdate (NiMoO4) using chitosan as a biopolymer. The cross-linked NiMoO4 showed an improved areal specific capacitance of 1250 mF cm−2 and 85% of its initial capacitance is retained after 2000 cycles. The morphology of the cross-linked NiMoO4 deduced using a field emission scanning microscope and a transmission electron microscope showed the formation of nanofibre-like dendrites having a porous network. The intramolecular interactions that facilitate the electron transfer path with more active sites for nucleation are attributed to higher capacitance. X-ray photoelectron spectroscopy measurements ascertain the presence of NiMoO4 in the aniline composite after the electropolymerization process. Overall, the surfactant exhibited surface functionality, whereas the biopolymer tuned the redox reactions of the NiMoO4 electrode.

 Please wait while we load your content...
Please wait while we load your content...