Effect of metal complexation on the equilibrium between methylphosphepine and methylphosphanorcaradiene and their benzo analogues†
Abstract
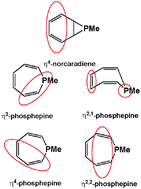
Theoretical studies are reported on methylphosphepine, methylbenzophosphepine, their norcaradiene isomers, and their metal complexes with Fe(CO)3 and CpCo (Cp = η5-C5H5) units. Both methylphosphepine and methylphosphanorcaradiene are C6H6PCH3 species existing as anti/syn stereoisomer pairs with the methylphosphepine structures at slightly higher energies. The transition states for the interconversion of these isomers and their benzo derivatives lie ∼20 kcal mol−1 in energy above the methylphosphanorcaradiene isomers. Complexation of either C6H6PCH3 ligand with the transition metal units Fe(CO)3 and CoCp leads to energetically closely spaced η4-tetrahapto and η3-trihapto isomers of the methylphosphepine complexes and η4-tetrahapto isomers of the methylphosphanorcaradiene complexes. However, the bis(dihapto) (η2,2-C6H6PCH3)Fe(CO)3 and (η2,2-C6H6PCH3)CoCp complexes involving coordination of non-adjacent C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bonds lie at significantly higher energies. Fusion of a benzene ring to the C6H6PCH3 rings in methylphosphepine and methylphosphanorcaradiene leads to significantly different structures of their lowest energy metal complexes. Thus the lowest energy (C10H8PCH3)Fe(CO)3 and (C10H8PCH3)CoCp structures are η2,2 and η4 methylbenzophosphepine complexes, which avoid using any carbon atoms of the benzene ring in the ligand for metal complexation. Higher energy (C10H8PCH3)Fe(CO)3 and (C10H8PCH3)CoCp structures have tetrahapto ligands with one or both C
C double bonds lie at significantly higher energies. Fusion of a benzene ring to the C6H6PCH3 rings in methylphosphepine and methylphosphanorcaradiene leads to significantly different structures of their lowest energy metal complexes. Thus the lowest energy (C10H8PCH3)Fe(CO)3 and (C10H8PCH3)CoCp structures are η2,2 and η4 methylbenzophosphepine complexes, which avoid using any carbon atoms of the benzene ring in the ligand for metal complexation. Higher energy (C10H8PCH3)Fe(CO)3 and (C10H8PCH3)CoCp structures have tetrahapto ligands with one or both C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bonds of the benzene ring complexed with the metal atom.
C double bonds of the benzene ring complexed with the metal atom.

 Please wait while we load your content...
Please wait while we load your content...