A recyclable Mn–porphyrin catalyst for enantioselective epoxidation of unfunctionalized olefins using molecular dioxygen†
Abstract
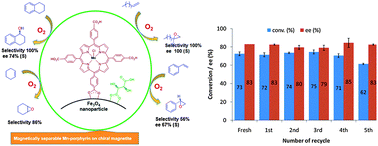
A magnetically separable chiral catalyst, Fe3O4/tart/Mn(TCPP)Cl, was prepared by the synthesis and immobilization of manganese–tetra(4-carboxyphenyl)porphyrin, Mn(TCPP)Cl, onto the chiral surface of magnetite nanoparticles (Fe3O4/tart-NPs; tart = L-tartaric acid) through the carboxylate groups. The solid catalyst was characterized by a variety of methods including XRD, FE-SEM, FT-IR and UV-Vis spectroscopy. The catalytic activity was tested in the epoxidation reactions of prochiral olefins with molecular oxygen. The reactions were carried out in acetonitrile as solvent with molecular oxygen in the presence of isobutyraldehyde as the stoichiometric oxidant. The catalyst showed consistently high conversion, selectivity and enantioselectivity to epoxide formation in the oxidation of a variety of terminal, cyclic and aromatic olefins. The catalyst showed little deactivation with time and it is easily recovered by magnetic filtration and could be reused four times with little or no loss in activity and selectivity.


 Please wait while we load your content...
Please wait while we load your content...