Electrochemical evaluation of ciprofloxacin adsorption on soil organic matter
Abstract
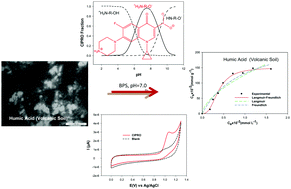
Over the last decade, at both a national and international level, the amount and fate of pharmaceutical residues in the environment have become of public interest. Among the several types of pharmaceutical wastes, a particular fluoroquinolone called ciprofloxacin (Cipro) is worth mentioning. On the other hand, soil organic matter (OM) has proven to be capable of generating interactions with antibiotics, which results in a decrease in their availability. Therefore, the main goal of the current work is to investigate the adsorption of Cipro on humic acids (HA), and to evaluate an electrochemical adsorption experimental design to handle the HA special particle size and solubility. S-type adsorption isotherms are observed for Cipro adsorption on HA extracted from two volcanic soils, wherein a cooperative adsorption phenomenon exists. The Langmuir–Freundlich model was utilised to obtain the best fit. This model is especially effective at fitting HA–Andisol adsorption, R2 > 0.99. Adsorption on both HA presents n > 1, strongly suggesting the existence of cooperative adsorption. This comes from the macromolecular nature of the multiple functional groups, which results in multiple interactions. The analytical results of the electrochemical methodology are statistically precise and accurate. The electrochemical technique requires shorter adsorbent–adsorbate equilibration times than traditional methods and physical separation is not needed. A detailed particle size analysis shows that the adsorption is the most significant process in OM–ciprofloxacin interaction. In relation to analytical parameters of pharmaceutical samples (ciprofloxacin), a relative standard deviation of 1.85%, a percentage error of 1.53% and a 98.5% of recovery were obtained.

 Please wait while we load your content...
Please wait while we load your content...