In situ formation of bromobenzene diazonium ions and their spontaneous reaction with carbon-coated LiFePO4 in organic media†
Abstract
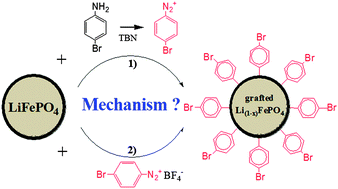
In this article, the spontaneous reaction, in organic media, of bromobenzene diazonium ions with carbon coated-LiFePO4 particles is reported. The formation of bromobenzene diazonium ions from the corresponding amine and tert-butyl nitrite was also investigated. The modified powders and the reaction mixture were analyzed to gain more insight into the grafting mechanism. The modified powders were characterized by thermogravimetric analyses to assess the loading of grafted molecules and inductively coupled plasma atomic emission spectrometry to estimate the number of moles of deinserted Li+ ions after reaction. Gas chromatography coupled with mass spectrometry measurements provided the yield of diazotization that was determined under various experimental conditions. It was demonstrated that the nature of the substrate strongly affected the yield of diazotization. Although LiFePO4/C is spontaneously reducing in situ generated bromobenzene diazonium ions to form the grafted layer, a significant fraction of diazonium ions is involved in side reactions. The oxidation of LiFePO4/C by tert-butyl nitrite is decreasing the diazotization yield when using LiFePO4/C as a substrate. The utilization of 4-bromobenzene diazonium tetrafluoroborate salt resulted in not only a slightly higher loading of grafted groups but also a higher oxidation of LiFePO4/C. The presence of by-products in solution after the grafting reaction also suggested that all species generated by the reduction of 4-bromobenzene diazonium ions are not forming a grafted layer. It has been shown that the grafting mechanism is very similar for in situ generated diazonium ions and diazonium salt with grafting efficiencies of about 10 and 14%, respectively.

 Please wait while we load your content...
Please wait while we load your content...