A one-pot sequential five-component domino reaction for the expedient synthesis of polysubstituted pyrroles†
Abstract
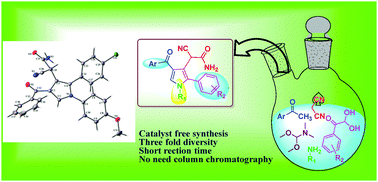
A series of novel pyrrole derivatives has been synthesized from readily available acetophenone, N,N-dimethylformamide dimethyl acetal, aniline, arylglyoxal and malononitrile in methanol under reflux conditions. This one-pot reaction constitutes a new and rapid route to the construction of a library of pyrroles. The synthesized compounds are characterized using spectral techniques and single crystal X-ray study.

 Please wait while we load your content...
Please wait while we load your content...