Monitoring thermo-reversible dehydration of the pluronic microenvironment using 4-chloro-1-naphthol as an ESPT fluorescent molecular probe†
Abstract
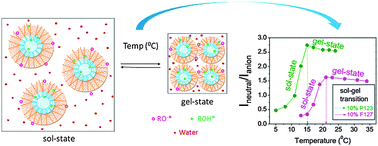
Pluronics are polymeric surfactants that undergo sol–gel phase transition as a function of both temperature and concentration and have a high impact on therapeutics. An excited state proton transfer (ESPT) fluorescent molecular probe, 4-chloro-1-naphthol, has been employed to explore the sol–gel transition of pluronics in aqueous media. In aqueous homogeneous media, 4-chloro-1-naphthol shows anionic emission, whereas in heterogeneous media, it results in dual emission of its neutral and anionic forms. Moreover, variations in the intensity (I) ratio, Ineutral/Ianion, and area (A) ratio under the two individual curves, Aneutral/Aanion, faithfully reflect the progressive changes in the medium heterogeneity. The values of both Ineutral/Ianion and Aneutral/Aanion increase with increasing temperature during sol–gel phase transition and reach maxima at the phase transition temperature. Moreover, the higher values of Ineutral/Ianion and Aneutral/Aanion in 10% P123 compared to those in F127 signify a lower micro-polarity of the P123 medium compared to that of F127. Evidence of progressive dehydration is obtained from the excited state decay dynamics. It shows that the proton transfer rate decreases remarkably up to the sol–gel transition temperature and after that it remains almost constant. In the present study, thermo-reversible sol–gel transition of pluronics (P123, F127) along with the dehydration of micelles has been monitored with 4-chloro-1-naphthol.

 Please wait while we load your content...
Please wait while we load your content...