Inclusion complexes of propiconazole nitrate with substituted β-cyclodextrins: the synthesis and in silico and in vitro assessment of their antifungal properties†
Abstract
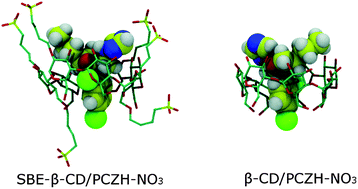
Inclusion complexes of protonated propiconazole nitrate (PCZH-NO3) with three substituted cyclodextrin (CD) derivatives, namely, sulfobutylether-β-CD (SBE7-β-CD), sulfated-β-CD (β-CD-SNa) and monochlorotriazinyl-β-CD (MCT-β-CD), were investigated as new antifungal systems and compared with a previously published complex of PCZH-NO3 with unsubstituted β-CD. The inclusion complexes were prepared by the freeze-drying method and were characterized by 1H-NMR, 2D Roesy NMR and DSC. This study demonstrates the coexistence of two types of PCZH-NO3 inclusion into the CD cavity. The complexes with SBE7-β-CD had the highest association constant values and the inclusion efficiency was close to 100%. Comparative in silico docking and molecular dynamics simulations were performed. The antifungal activity was assessed on Candida spp. and was similar for all four inclusion complexes. The cytotoxicity assessed on normal human dermal fibroblasts (NHDF) was significantly higher for the complex with β-CD than that of the complexes of substituted CDs.

 Please wait while we load your content...
Please wait while we load your content...