One-dimensional assembly of functional proteins: toward the design of an artificial cellulosome†
Abstract
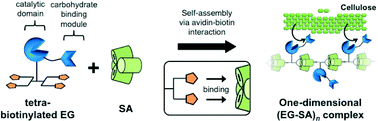
In biological systems, proteins can form well-organized, higher-order structures with unique functions that would be difficult to achieve with a single protein. These proteinaceous supramolecular structures form by self-assembly, and the spatial arrangement of the protein building blocks in them is very important. In the present study, an artificial system was developed using recombinant proteins as building blocks, which were assembled in a one-dimensional manner. The assembly of these building blocks was based on the avidin–biotin interaction. A tetrameric biotin ligand unit was designed so that the 1 : 4 stoichiometry of the avidin–biotin interaction was altered to a 1 : 2 directional interaction between the streptavidin and tetrabiotinylated protein units. In a proof-of-concept study, site-specifically tetrabiotin-labeled endoglucanase and cellulose-binding module units were prepared, and then these components were self-assembled by mixing with streptavidin to mimic a natural cellulosome. The formation of one-dimensional assemblies of the protein units depended on the stoichiometry of the avidin–biotin interaction sites in the system. Interestingly, the saccharification efficiency improved when the component ratio of protein units in the assemblies was changed. The presence of the optimal ratio of the building blocks implies the modularity of the present protein assembly system.

 Please wait while we load your content...
Please wait while we load your content...