Microbial cyclosophoraose as a catalyst for the synthesis of diversified indolyl 4H-chromenes via one-pot three component reactions in water†
Abstract
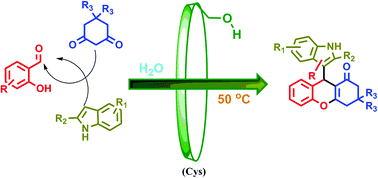
As a novel biosourced saccharide catalyst, microbial cyclosophoraose, a cyclic β-(1,2) glucan, was used for the synthesis of therapeutically important versatile indolyl 4H-chromenes via a one pot three-component Knoevenagel–Michael addition–cyclization reaction of salicylaldehyde, 1,3-cyclohexanedione/dimedone, and indoles in water under neutral conditions. A possible reaction mechanism through molecular complexation is suggested based on 2D ROESY NMR spectroscopic analysis. Moreover, green chemistry metric calculations were carried out for a model reaction, indicating the satisfactory greener approach of this method, with a low E-factor (0.18) and high atom economy (AE = 91.20%). The key features of this protocol are based on two critical factors where the first is to use a novel eco-friendly supramolecular carbohydrate catalyst and the second is its fine green properties such as compatibility with various substituted reactants, recyclability of the catalyst, chromatography-free purification, high product selectivity, and clean conversion with moderate to excellent yields in an aqueous medium.

 Please wait while we load your content...
Please wait while we load your content...