Structural and chemical evolution of methylammonium lead halide perovskites during thermal processing from solution†
Abstract
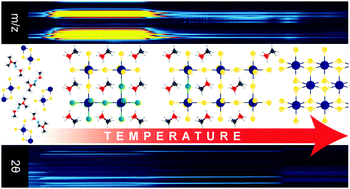
Following the prominent success of CH3NH3PbI3 in photovoltaics and other optoelectronic applications, focus has been placed on better understanding perovskite crystallization from precursor and intermediate phases in order to facilitate improved crystallinity often desirable for advancing optoelectronic properties. Understanding of stability and degradation is also of critical importance as these materials seek commercial applications. In this study, we investigate the evolution of perovskites formed from targeted precursor chemistries by correlating in situ temperature-dependent X-ray diffraction, thermogravimetric analysis, and mass spectral analysis of the evolved species. This suite of analyses reveals important precursor composition-induced variations in the processes underpinning perovskite formation and degradation. The addition of Cl− leads to widely different precursor evolution and perovskite formation kinetics, and results in significant changes to the degradation mechanism, including suppression of crystalline PbI2 formation and modification of the thermal stability of the perovskite phase. This work highlights the role of perovskite precursor chemistry in both its formation and degradation.

 Please wait while we load your content...
Please wait while we load your content...