A CO2 adsorption-enhanced semiconductor/metal-complex hybrid photoelectrocatalytic interface for efficient formate production†
Abstract
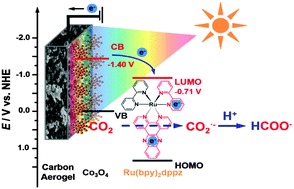
In photoelectrochemical CO2 conversion, the concentration of fixed CO2 on the photocathode surface is of primary concern. Herein, a CO2 adsorption-enhanced semiconductor/metal-complex hybrid photoelectrocatalytic interface was established by utilizing a carbon aerogel as the CO2 fixation substrate. In CO2 reduction photoelectrocatalysis, Co3O4 was employed as the light harvester, and Ru(bpy)2dppz was utilized as the electron transfer mediator and CO2 activator. The CO2 surface concentration exhibited a 380-fold increase on this hybrid interface than that on Co3O4/FTO. The CO2 conversion to formate occurred at an onset potential of −0.45 V (vs. normal hydrogen electrode, NHE) under photoelectrochemical conditions, 160 mV more positive than its thermodynamic redox potential. At an applied potential of −0.60 V (vs. NHE), the selectivity of the formate yield reached 99.95%, with a production rate of approximately 110 μmol cm−2 h−1 and a Faradaic efficiency of 86%. Such a conversion has an electron transfer rate of 2.94 × 10−3 cm s−1. The CO2 conversion to formate was confirmed to be an instantaneous proton-coupled electron transfer process, originating from the rapid photoelectrochemical activation of bpy and dppz in Ru(bpy)2dppz as well as the synergic effect of the promoted CO2 adsorption and the applied molecular catalysis.

 Please wait while we load your content...
Please wait while we load your content...