Solvent-induced structural diversity in tetranuclear Ni(ii) Schiff-base complexes: the first Ni4 single-molecule magnet with a defective dicubane-like topology†
Abstract
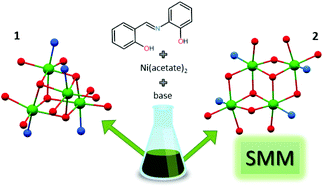
Two tetranuclear NiII complexes, namely [Ni4(L)4(CH3OH)3(H2O)]·CH3OH (1) and (Pr3NH)2[Ni4(L)4(CH3COO)2] (2, Pr3N = tripropylamine), were synthesized from a tridentate Schiff base ligand H2L (2-[(E)-(2-hydroxybenzylidene)amino]phenol) and Ni(CH3COO)2·4H2O, using different solvents and their ratios (CH3OH and/or CH2Cl2). The prepared Ni4 complexes are of different structural types, involving an Ni4O4 cubane-like core (1) and Ni4O6 defective dicubane-like core (2), with all the Ni atoms hexacoordinated. The complexes were characterized by elemental analysis, FT-IR spectroscopy, variable temperature and field magnetic measurements, and single crystal X-ray analysis. The DFT and CASSCF/NEVPT2 theoretical calculations were utilized to reveal information about the isotropic exchange parameters (Jij) and single-ion zero-field splitting parameters (Di, Ei). The variable temperature magnetic data suggested the competition of the antiferromagnetic and ferromagnetic intracluster interactions in compound 1, which is in contrast to compound 2, where all intracluster interactions are ferromagnetic resulting in the ground spin state S = 4 with an easy-axis type of anisotropy quantified by the axial zero-field splitting parameter D = −0.81 cm−1. This resulted in the observation of a field-induced slow-relaxation of magnetization (U = 3.3–6.7 K), which means that the complex 2 represents the first Ni4 single-molecule magnet with the defective dicubane-like topology.


 Please wait while we load your content...
Please wait while we load your content...