Effect of ligand denticity on the nitric oxide reactivity of cobalt(ii) complexes†
Abstract
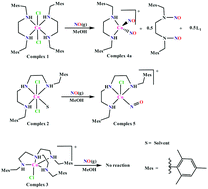
The activation of nitric oxide (NO) by transition metal complexes has attracted a wide range of research activity. To study the role of ligand denticity on the NO reactivity of Co(II) complexes, three complexes (1, 2 and 3) were prepared with ligands L1, L2 and L3 [L1 = N1,N2-bis(2,4,6-trimethylbenzyl)ethane-1,2-diamine; L2 = N1-(2,4,6-trimethylbenzyl)-N2-(2-((2,4,6-trimethylbenzyl)amino)ethyl)ethane-1,2-diamine] and L3 = N1-(2,4,6-trimethylbenzyl)-N2,N2-bis(2-((2,4,6-trimethylbenzyl)amino)ethyl)ethane-1,2-diamine], respectively. The complexes differ from each other in terms of denticity and flexibility of the ligand frameworks. In degassed methanol solution, they were exposed to NO gas and their reactivity was studied using various spectroscopic techniques. In the case of complex 1 with a bidentate ligand, reductive nitrosylation of the metal ion with concomitant dinitrosation of the ligand framework was observed. Complex 2 with a tridentate ligand did not undergo reductive nitrosylation; rather, the formation of [CoIII(NO−)] was observed. The nitrosyl complexes were isolated and structurally characterized. On the other hand, complex 3 with a tetradentate tripodal ligand did not react with NO. This can be attributed to the geometry of the complex as well as due to the accessibility of the corresponding redox potential.

 Please wait while we load your content...
Please wait while we load your content...