Superior catalytic activity of synthesized triangular silver nanoplates with optimized sizes and shapes
Abstract
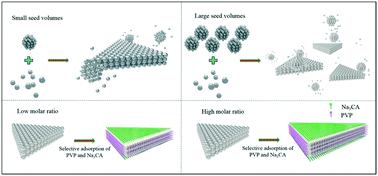
Triangular silver nanoplates (Ag-NPs) were synthesized using a rapid and reproducible chemical reduction method involving both seeded growth and the selective adhesion of capping ligands. The average edge length of the nanoplates and the position of the plasmon resonance band could be tuned over a wide range by simply adjusting the experimental conditions. Using the appropriate seed volume in the growth solution and molar ratio of the capping ligand PVP to the precursor AgNO3 were found to be indispensable for forming uniform silver nanoplates. Specifically, testing the molar ratio of PVP to AgNO3 yielded thin nanoplates with edge lengths up to 310 nm, and hence a high aspect ratio. Nanoplates prepared with different seed volumes and PVP-to-AgNO3 molar ratios were structurally characterized. In addition, the changes in the edge length and size of the Ag nanoplates were studied. The main UV-visible absorption peak of the nanoplate solution showed a blue shift of the surface plasmon band when the seed volume was increased, and a red shift when the molar ratio was increased. The catalytic performance of the synthesized Ag-NPs toward the reduction of the dye rhodamine B (Rh B) by NaBH4 was established, and the degradation reaction was observed to follow first-order kinetics.


 Please wait while we load your content...
Please wait while we load your content...