A green-by-design system for efficient bio-oxidation of an unnatural hexapyranose into chiral lactone for building statin side-chains†
Abstract
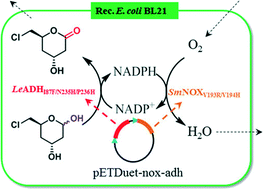
A novel NADP+-dependent aldehyde dehydrogenase (LeADH) was identified from Lodderomyces elongisporus and engineered for bio-oxidation of (3R,5S)-6-chloro-2,4,6-trideoxy-erythro-hexapyranose, a key chiral intermediate, for synthesis of statin side-chains. The catalytic activity of LeADH was improved by 12-fold through the combinatorial active-site saturation test (CAST) strategy. The resultant variant, LeADHI87F/N235H/P236H, was co-expressed with an NADPH oxidase SmNOXV193R/V194H in Escherichia coli. A green-by-design system was then constructed for efficient oxidation of the unnatural hexapyranose into the key statin side chain, (3R,5S)-6-chloro-2,4,6-trideoxy-erythro-hexonolactone, with oxygen as the co-substrate and water as the sole by-product.

 Please wait while we load your content...
Please wait while we load your content...