Critical role of interfacial effects on the reactivity of semiconductor-cocatalyst junctions for photocatalytic oxygen evolution from water†
Abstract
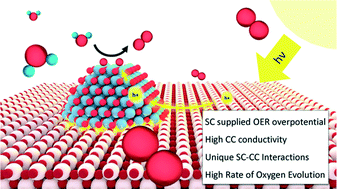
Photocatalytic water splitting has the potential to provide a sustainable approach for storing the energy of solar photons in the form of chemical bonds. Much work has been executed towards developing semiconducting light absorbers (SC) and co-catalysts (CC) for photoelectrochemical (PEC) and particulate photocatalytic (PPC) water splitting systems. However, minimal insights exist into how the formation of junctions between highly dispersed CC clusters and nanoparticle SCs influences the reactivity of the system, as compared to the reactivity of the CCs in electrochemical environments. In this work, 15 materials consisting of five different CC nanoclusters deposited on three different nanoparticle SCs were synthesized, characterized, and tested for oxygen evolution reaction (OER) activity. The SCs were chosen based on their similar n-type behavior and low OER activity, and the CCs were chosen based on their benchmarked OER reactivity in electrochemical measurements. We found that the photocatalytic activity of these samples for the OER did not directly correlate with the reactivity of the CCs measured under electrochemical conditions. Instead, the performance was controlled by interfacial effects specific to each SC|CC junction. CC electrical conductivity and SC supplied OER overpotential were the major factors identified to strongly affect OER rate. This work suggests that when designing an optimal photocatalyst, one can not simply deposit the most active CC (as measured in electrochemical conditions) on the SC which absorbs the highest fraction of the solar flux, as the CC–SC pairing requires critical interfacial considerations.
- This article is part of the themed collection: Nanocatalysis

 Please wait while we load your content...
Please wait while we load your content...