(Pyridyl)benzoazole palladium(ii) complexes as homogeneous catalysts in hydrogenation of alkenes and alkynes†
Abstract
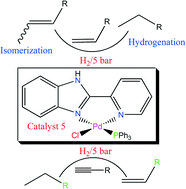
Reactions of 2-(2-pyridyl)benzimidazole (L1), 2-(2-pyridyl)benzothiazole (L2) and 2-(2-pyridyl)benzoxazole (L3) with either [PdCl2(NCMe)2] or [PdClMe(COD)] produced complexes [(PdCl2(L1)] (1), [(PdCl2(L2)] (2), [(PdCl2(L3)] (3) and [(PdClMe(L1)] (4) in good yields. Treatment of 1 with PPh3 gave the cationic complex [(Pd(L1)ClPPh3]Cl (5), while reactions of 4 with one equiv. of PPh3 and NaBAr4 (Ar = 3,5-(CF3)2C6H3) produced the cationic complex [(Pd(L1)MePPh3]BAr4 (6). Single-crystal X-ray analysis has been used to elucidate the structure of complex 6. The complexes formed active catalysts in hydrogenation reactions of alkenes and alkynes. Hydrogenation of terminal alkenes was accompanied by isomerization to the internal isomers, while alkyne reactions involved a two-step process producing alkenes and the respective alkanes. While the kinetics data and formation of inactive palladium nanoparticles support the homogeneous nature of the active species, mercury drop experiments indicated a possible role of the nanoparticles in the re-generation of the active species.

 Please wait while we load your content...
Please wait while we load your content...