Effects of tetrahedral molybdenum oxide species and MoOx domains on the selective oxidation of dimethyl ether under mild conditions†
Abstract
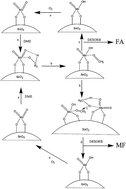
A new preparation method for MoO3–SnO2 catalysts precipitated by HNO3 was developed to selectively synthesize industrially useful chemicals formaldehyde and methyl formate via oxidation of environmentally friendly feedstock dimethyl ether. By adjusting the structure of MoO3–SnO2, the selectivity to formaldehyde and methyl formate can reach as high as 95.0% and 82.4%, respectively, under different conditions. The conclusion has been drawn, based on the experimental results, that the formation of tetrahedral molybdenum oxide species and SnMoO4 species favors the reaction of dimethyl ether to formaldehyde, and that the formation of MoOx domains favors the direct oxidation reaction of dimethyl ether to formaldehyde. The XRD and Raman results suggest that the formation of tetrahedral molybdenum oxide species has a positive correlation with the high selectivity to formaldehyde and indicates that the formation of the MoOx domains favors the high selectivity to methyl formate. The XPS results of the MoO3–SnO2 catalysts with different SnO2 contents demonstrate that formaldehyde is not readily desorbed from Mo1Sn2 and Mo1Sn3 because the electron-poor Mo cations of the domains supported on the surface of the catalysts strengthen their affinity for binding electron-donating dimethyl ether-derived intermediates and formaldehyde, which favors the subsequent reaction of formaldehyde to methyl formate. These interesting findings reveal that tetrahedral molybdenum oxide species and MoOx domains play an important role in the selective oxidation of dimethyl ether to formaldehyde and methyl formate, and give insight into the rational design of the MoO3–SnO2 catalyst structure for dimethyl ether oxidation in future studies.

 Please wait while we load your content...
Please wait while we load your content...