Computational insights into the reaction mechanism of methanol-to-olefins conversion in H-ZSM-5: nature of hydrocarbon pool†
Abstract
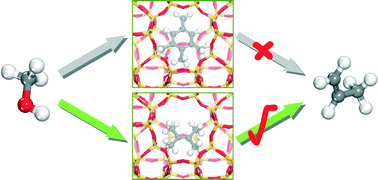
The nature of active hydrocarbon pool (HP) species for the methanol-to-olefins (MTO) conversion in zeolite catalysis still remains controversial. In this work, the catalytic cycles in which aromatics and olefins act as the HP species were investigated in H-ZSM-5 using periodic density functional theory calculations. Distribution of polymethylbenzenes (MBs) was qualitatively evaluated using static adsorption and dynamic interconversion analysis. It is revealed that 1,3,5-trimethylbenzene exhibits the strongest adsorption while 1,2,3,5-tetramethylbenzene (TMB) is the primary component of MBs in H-ZSM-5. The aromatic-based side chain cycle was found to be kinetically more demanding using p-xylene, 1,2,3,5-TMB, and 1,2,4,5-TMB as HP species to propagate ethyl side chain. The olefin-based cycle was illustrated using 2,3-dimethyl-2-butene (iso-C6) as HP species in a way similar to the aromatic-based side chain cycle, enabling the direct comparison of the overall kinetics. The involved methylation and cracking steps in the iso-C6-based cycle are more facile than those of the aromatic-based side chain cycle in H-ZSM-5. The aromatic-based paring cycle can also be excluded as the involved intermediate ions with five-membered rings are extremely unstable. As a result, it is most likely that olefins themselves rather than the aromatics serve as the HP species for the MTO conversion in H-ZSM-5.

 Please wait while we load your content...
Please wait while we load your content...