Electron transfer across the germanium–polyelectrolyte–gold nanoparticle interface: convenient detection and applications in sensing†
Abstract
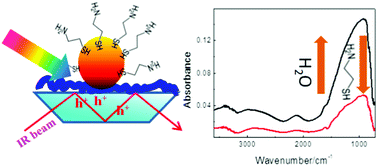
Germanium–polyelectrolyte–gold nanoparticle composites were prepared and characterized using FTIR-ATR spectroscopy and scanning electron microscopy. The germanium (Ge) element served as an internal reflection element and the buildup of the layered system was followed in situ. A positively charged polyelectrolyte, poly (allylamine hydrochloride) (PAH) adsorbs spontaneously on negatively charged Ge. Citrate-stabilized gold nanoparticles can then be adsorbed onto the PAH layer. Upon illumination of the device with visible light a prominent absorption over the entire mid infrared region is observed which is due to intervalence band transitions in Ge. The strong infrared signals are evidence for holes in the valence band of the Ge semiconductor, which arise due to electron transfer to the gold nanoparticles (GNPs). The electron transfer, as evidenced by the holes in Ge, is affected by the nature of the gap between the Ge semiconductor and the GNPs. Increasing the gap by adsorbing polyelectrolyte multilayers hinders the electron transfer. Also, heating and vacuum have a pronounced effect. The device is proposed as a sensor, where the sensing event is transduced into an optical signal in the infrared region, as demonstrated for a thiol molecule. The thiol has a large affinity for gold, and therefore affects the germanium–gold nanoparticle gap. This reduces the electron transfer, and therefore the absorption in the infrared region upon illumination with visible light. Removal of the thiol from the solution leads to the recovery of the signal.

 Please wait while we load your content...
Please wait while we load your content...