Exploring the mechanism of F282L mutation-caused constitutive activity of GPCR by a computational study
Abstract
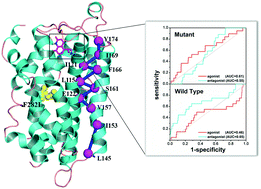
G-protein-coupled receptors (GPCRs) are important drug targets and generally activated by ligands. However, some experiments found that GPCRs also give rise to constitutive activity through some mutations (viz., CAM), which are usually associated with different kinds of diseases. However, the mechanisms of CAMs and their roles in interactions with drug-ligands are unclear in experiments. Herein, we used microsecond molecular dynamics simulations to study the effect of one important F282L mutation on β2AR in order to address the questions above. With the aid of principle component and correlation analysis, our results revealed that the F282L mutation could increase the instability of the overall structure, increase the dramatic fluctuations of NPxxY and extracellular loops, and decrease restraint of the helices through weakening interhelical H-bonding and correlations between residues, which could partly contribute to the constitutive activity reported by the experiments. The observations from the protein structure network (PSN) analysis indicate that the mutant exhibits less information flow than the wild β2AR and weakens the role of TM5 and TM6 in the signal transmission, but it enhances the impact of TM3 on the orthosteric pathway and TM4 on the allosteric one. In addition, the results from the virtual screening reveal that the mutant prefers to select agonists rather than antagonists, similar to the active state but opposite of the inactive state, further confirming that the F282L mutation advances the activation of β2AR. Our observations provide valuable information for understanding the mechanism of the mutation-caused constitutive activity of GPCR and related drug-design.

 Please wait while we load your content...
Please wait while we load your content...