Quantification and classification of substituent effects in organic chemistry: a theoretical molecular electrostatic potential study†‡
Abstract
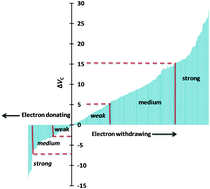
Substituent effects in organic chemistry are generally described in terms of experimentally derived Hammett parameters whereas a convenient theoretical tool to study these effects in π-conjugated molecular systems is molecular electrostatic potential (MESP) analysis. The present study shows that the difference between MESP at the nucleus of the para carbon of substituted benzene and a carbon atom in benzene, designated as ΔVC, is very useful to quantify and classify substituent effects. On the basis of positive and negative ΔVC values, a broad classification of around 381 substituents into electron withdrawing and donating categories is made. Each category is again sorted based on the magnitude of ΔVC into subcategories such as very strong, strong, medium, and weak electron donating/withdrawing. Furthermore, the data are used to show the transferability and additivity of substituent effects in π-conjugated organic molecules such as condensed aromatic, olefinic, acetylenic, and heterocyclic systems. The transferability properties hold good for ΔVC in all these molecular systems. The additive properties of substituent effects are strongly reflected on ΔVC and the predictive power of the data to assign the total substituent effects of multi-substituted systems is verified. The ΔVC data and the present classification of substituents are very useful to design π-conjugated organic molecular systems with desired electron rich/poor character.

 Please wait while we load your content...
Please wait while we load your content...