Specific ion modulated thermoresponse of poly(N-isopropylacrylamide) brushes†
Abstract
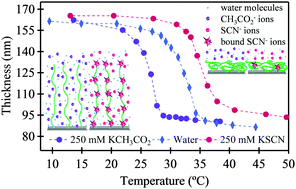
The influence of specific anions on the equilibrium thermoresponse of poly(N-isopropylacrylamide) (pNIPAM) brushes has been studied using in situ ellipsometry, quartz crystal microbalance with dissipation (QCM-D) and static contact angle measurements between 20 and 45 °C in the presence of up to 250 mM acetate and thiocyanate anions in water. The thickness and changes in dissipation exhibited a broad swelling transition spanning approximately 15 °C from collapsed (high temperatures) to swollen conformation (low temperatures) while the brush surface wettability changed over approximately 2 °C. In the presence of the kosmotropic acetate anions, the measured lower critical solution temperature (LCST) by the three techniques was very similar and decreased linearly as a function of ionic strength. Conversely, increasing the concentration of the chaotropic thiocyanate anions raised the LCST of the pNIPAM brushes with variation in the measured LCST between the three techniques increasing with ionic strength. The thickness of the pNIPAM brush was seen to progressively increase with increasing thiocyanate concentration at all temperatures. It is proposed that specific ion binding of the chaotropic thiocyanate anion with pNIPAM amide moieties increases the electrostatic intra- and intermolecular repulsion within and between pNIPAM chains. This allows the brush to begin to swell at higher temperatures and to an overall greater extent.


 Please wait while we load your content...
Please wait while we load your content...