Electrochemical studies of hydrogen chloride gas in several room temperature ionic liquids: mechanism and sensing†
Abstract
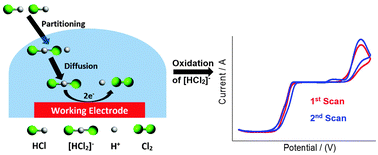
The electrochemical behaviour of highly toxic hydrogen chloride (HCl) gas has been investigated in six room temperature ionic liquids (RTILs) containing imidazolium/pyrrolidinium cations and range of anions on a Pt microelectrode using cyclic voltammetry (CV). HCl gas exists in a dissociated form of H+ and [HCl2]− in RTILs. A peak corresponding to the oxidation of [HCl2]− was observed, resulting in the formation of Cl2 and H+. These species were reversibly reduced to H2 and Cl−, respectively, on the cathodic CV scan. The H+ reduction peak is also present initially when scanned only in the cathodic direction. In the RTILs with a tetrafluoroborate or hexafluorophosphate anion, CVs indicated a reaction of the RTIL with the analyte/electrogenerated products, suggesting that these RTILs might not be suitable solvents for the detection of HCl gas. This was supported by NMR spectroscopy experiments, which showed that the hexafluorophosphate ionic liquid underwent structural changes after HCl gas electrochemical experiments. The analytical utility was then studied in 1-ethyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide ([C2mim][NTf2]) by utilising both peaks (oxidation of [HCl2]− and reduction of protons) and linear calibration graphs for current vs. concentration for the two processes were obtained. The reactive behaviour of some ionic liquids clearly shows that the choice of the ionic liquid is very important if employing RTILs as solvents for HCl gas detection.

 Please wait while we load your content...
Please wait while we load your content...