A series of coordination polymers with tuned terphenyl tetracarboxylates and bis-pyridyl ligands with different flexibilities manifesting fluorescence properties and photocatalytic activities†
Abstract
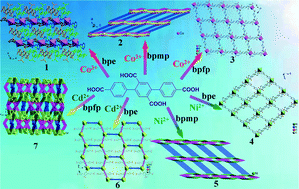
A family of new two-dimensional (2D) and three-dimensional (3D) coordination polymers, namely, [Co(H2L)(bpe)2]n (1), [Co2(L)(bpmp)(H2O)]n (2), [Co(H2L)(bpfp)(H2O)2]n (3), {[Ni(H2L)(bpe)(H2O)2]·2H2O}n (4), [Ni(L)0.5(bpmp)0.5]n (5), {[Cd(H2L)(bpe)0.5(H2O)2]·0.5(bpe)}n (6) and {[Cd2(L)(bpfp)0.5]·H2O}n (7) was constructed using [1,1′ : 4′,1′′-terphenyl]-2′,4,4′′,5′-tetracarboxylic acid (H4L), the transition metals and the bis-pyridyl ligands (bpe = 1,2-bis(pyridin-4-yl)ethene, bpmp = N,N′-bis(4-pyridyl)piperazine and bpfp = bis(4-pyridylformyl)piperazine) with different flexibilities. These complexes were characterized by elemental analyses, IR spectra, thermogravimetric (TG) analyses, powder X-ray diffraction (PXRD) and single-crystal X-ray diffraction. Complex 1 comprises a 2D coordinated layer that consists of CoII metal ions and H2L ligands, and monodentate bpe ligands extend on both sides of the network. Complex 2 is a (4,6)-connected 3D framework based on Co-L 2D sheets with four types of different quadrilaterals and bridging bpmp ligands. When bpmp is replaced by bpfp, complex 3 possesses a 2D (4,4) network containing “H”-type 1D Co-H2L chains and 1D Co-bpfp zigzag chains. Complex 4 is similar to complex 3 except that it contains 1D Ni-bpe linear chains. Complex 5 displays a 3D framework with symmetrical polygonal windows, in which there are 2D grids composed of quadrilaterals of the same size. Complex 6 exhibits a 2D {63} layer including hexagonal grids, and some non-coordinated bpe ligands are present in the network. When bpfp is used in complex 7, a complicated 3D {415·613}2{42·63·8}{47·63}4 framework is formed. In this structure, all the coordinated atoms from L and bpfp ligands coordinate with CdII metal ions. Furthermore, the 2D layers of complexes 1, 3, 4 and 6 are ultimately expanded into 3D supramolecular frameworks via hydrogen-bonding interactions. The flexibility of the bis-pyridyl ligands indicates a consequential influence on the final architectures of complexes 1–7. Moreover, their electrochemical behaviors and fluorescence properties have been investigated. Photocatalytic properties reveal that complex 3 exhibits good catalytic activities for the degradation of the organic dye methylene blue (MB).

 Please wait while we load your content...
Please wait while we load your content...