Crystals on the move: mechanical effects in dynamic solids
Abstract
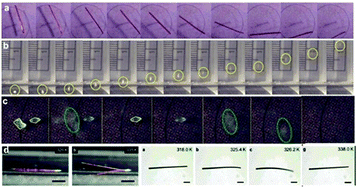
When exposed to external stimuli such as heat or light, certain single crystals can acquire momentum and undergo motion. On a molecular scale, the motility of such dynamic single crystals is triggered by a phase transition or chemical reaction without gaseous products, and macroscopically manifests as either slow (reversible or irreversible) deformation, or as rapid, almost instantaneous propulsion of the crystals that is oftentimes accompanied by disintegration. While the elastic energy of the slow reconfiguration processes such as bending, twisting and coiling can be utilized for actuation of other objects, the fast disintegrative processes could be exploited to initiate pressure-sensitive applications. This short review intends to summarize recent developments in the growing research on dynamic crystals, especially aspects of the mechanism of rapid motion of thermosalient and photosalient (leaping) crystals. The collective evidence indicates that these solids are organic-based analogues of the inorganic martensitic materials. While qualitative explanation of the molecular processes that lead to the related dynamic phenomena can be provided, quantification of their kinematics, estimation of the useful work that can be extracted, and prediction of their occurrence are yet to be established. Harnessing the potential of these materials to rapidly and efficiently perform the fundamentally important process of transduction of heat or light into kinetic energy appears as a prospective basis for their application in motion gears and devices.

 Please wait while we load your content...
Please wait while we load your content...