Genetically encoded fluorophenylalanines enable insights into the recognition of lysine trimethylation by an epigenetic reader†
Abstract
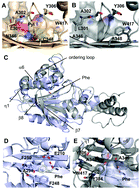
Fluorophenylalanines bearing 2–5 fluorine atoms at the phenyl ring have been genetically encoded by amber codon. Replacement of F59, a phenylalanine residue that is directly involved in interactions with trimethylated K9 of histone H3, in the Mpp8 chromodomain recombinantly with fluorophenylalanines significantly impairs the binding to a K9-trimethylated H3 peptide.

 Please wait while we load your content...
Please wait while we load your content...