Bifunctional tertiary amine-squaramide catalyzed asymmetric catalytic 1,6-conjugate addition/aromatization of para-quinone methides with oxindoles†
Abstract
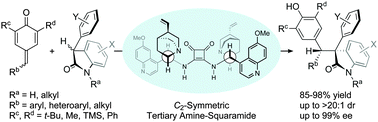
The asymmetric catalytic 1,6-addition of p-QMs with racemic oxindoles under the bifunctional catalysis of C2-symmetric dimeric Cinchona-derived squaramide is described. This tertiary amine-squaramide catalyzed reaction provides a diastereoselective and enantioselective approach to the effective assembly of diverse diarylmethine-substituted oxindoles having vicinal tertiary and quaternary stereocenters.

 Please wait while we load your content...
Please wait while we load your content...