Development of a new diagnostic Raman method for monitoring epigenetic modifications in the cancer cells of human breast tissue
Abstract
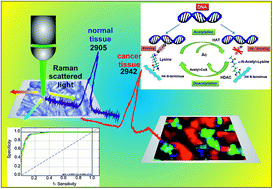
Cancer diagnosis requires better screening of early stages of pathology and monitoring patient responses to treatment. Current technologies in the clinical sector are expensive, sophisticated and time consuming. This paper develops a novel Raman based alternative for currently existing epigenetics research approaches. The proposed Raman approach can ‘upgrade’ cancer epigenetic tests and answer many questions by monitoring the biochemistry of cancer cells. We will show that Raman spectroscopy and Raman imaging can detect the relative amounts of acetylated and methylated lysine by monitoring the vibrations of the acetyl and methyl chemical functional groups. In comparison to existing tests and assays used to monitor molecular processes of acetylation and methylation of all proteins, Raman-based methods have the potential to be a powerful alternative for conventional methods of cell biology, because they allow non-invasive detection of cellular acetylation and methylation processes that are not limited to only those events that are sensitive to a specific antibody. Vibrational Raman signatures of acetylation and methylation processes in epithelial cells of human breast tissue (ductal and lobular carcinoma) have been used to identify and discriminate structures in normal and cancerous tissues. Our results demonstrate that the stretching vibration of the acetyl group observed at around 2938–2942 cm−1 and the methyl group at 2970 cm−1 by Raman spectroscopy is useful in monitoring these epigenetic molecular processes in cancer cells. The Raman vibrational marker is markedly blue-shifted from 2905 cm−1 (non-acetylated C–H vibrations) for normal cells to 2942 cm−1 (vibrations of the acetylated functional group) in cancer cells. The sensitivity and specificity obtained directly from PLSDA and cross-validation for a chosen model gives a sensitivity and specificity of 86.1% and 91.3% for calibration and 85.3% and 91.3% for cross-validation, respectively. The results presented in the paper provide strong evidence that the global acetylation level of histone and non-histone proteins increases in human breast cancer cells.

 Please wait while we load your content...
Please wait while we load your content...