A label-free fluorescence assay for thrombin activity analysis based on fluorescent protein and gold nanoparticles†
Abstract
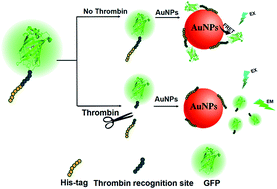
Thrombin, a kind of serine protease, is a significant regulator of thrombosis in vivo. Herein, we proposed a label-free and sensitive fluorescence assay for probing thrombin activity based on an enhanced green fluorescent protein (EGFP) probe and gold nanoparticles (AuNPs). The EGFP probe containing a hexahistidine sequence (His-tag) and a thrombin recognition site at the N-terminal was designed. His-tag enables the probe to be adsorbed on the surface of AuNPs through high affinity of His–Au bonds, and as a consequence, fluorescence resonance energy transfer (FRET) would happen, where EGFP is the fluorescence donor and AuNPs are the acceptors. Because of the high extinction coefficient of AuNPs, the fluorescence of EGFP will be quenched. When there is thrombin, it cleaves the recognition site, and results in the leaving of the His-tag from EGFP. EGFP without the His-tag cannot be efficiently adhered on AuNPs, and its fluorescence is protected. Thus, the corresponding fluorescence signal can respond to the activity of thrombin. Using this turn-on fluorescence assay, we obtained a highly sensitive and specific detection of thrombin activity (limit of detection = 0.0025 U mL−1) with a linear range from 0.01 U mL−1 to 0.25 U mL−1 under optimized conditions. The assay can also be used to measure the inhibition of thrombin activity (where hirudin was used as an example) with an IC50 value of 1.38 nM, and this was successfully performed on real samples. Furthermore, the proposed assay has the potential to be a fluorescence-colorimetry dual detection method.

 Please wait while we load your content...
Please wait while we load your content...