SERS speciation of the electrochemical oxidation–reduction of riboflavin†
Abstract
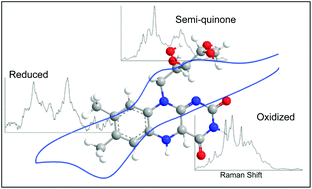
The reduction and oxidation of the flavin system is an important electron transfer reaction in biological systems. Several reaction pathways exist to connect oxidized to fully reduced riboflavin, each with unique intermediates including a semi-quinone radical. By performing surface-enhanced Raman scattering (SERS) with simultaneous electrochemical detection of riboflavin at different pH values, we are able to correlate reversible changes in spectral features to the current changes observed in the cyclic voltammetry. Multivariate curve resolution analysis of the SERS spectra indicates that three distinct components were present at the SERS electrode at each pH during the potential sweep. To verify and better understand the variations in Raman bands across the voltammogram, density functional theory (DFT) calculations were performed to model the effect of pH and oxidation state on the riboflavin Raman spectrum. The calculated spectra show qualitative agreement with the species identified in the chemometric analysis. This combination of results indicates the presence of the oxidized, semi-quinone, and reduced forms of riboflavin and provides insight into the mechanism of the flavin redox system.
- This article is part of the themed collection: Surface-enhanced Raman scattering

 Please wait while we load your content...
Please wait while we load your content...