Investigating non-specific binding to chemically engineered sensor surfaces using liposomes as models†
Abstract
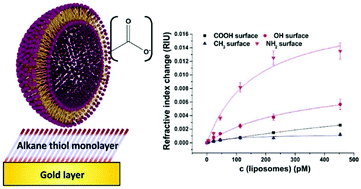
Nanoparticles are ubiquitously used for signal enhancement in (bio)sensors, but their true possible performance is typically hampered by non-specific binding. A better understanding of the nature and the prevention of non-specific binding through surface engineering of the particles and sensor surfaces is needed to intelligently design (bio)sensors and potentially avoid bulk blocking methods. Hence, two types of liposomes were used as model for signal-enhancing nanoparticles. Their surface was engineered to bear negative surface charge. One type was synthesized with additional 6 mol% –COOH groups. Their interaction with four typical chemically modified sensor surfaces was then mechanistically characterized by surface plasmon resonance (SPR) spectroscopy. It was shown that the non-specific binding can be described with Langmuir isotherms providing quantitative information of dissociation constants and surface loading with especially high correlation coefficients (>0.97) for all the studied sensor surfaces modified with hydrophilic alkane thiols. By tailoring the sensor surface chemistry, non-specific binding was significantly minimized. Here, carboxyl- or methyl-terminated surfaces performed best. In fact, the pairing of –COOH groups on the sensor surface with –COOH groups on the liposomes almost completely eliminated non-specific binding, resulting in a SPR signal change of only 1 mRIU (refractive index unit) at 100 μM phospholipid concentration. Surprisingly though, –OH groups on the surface, which are also commonly used in sensing applications, did not lead to decreased adsorption, but caused significant signal changes (4 mRIU at 100 μM phospholipid) due to non-specific binding. Overall, the mechanistic studies presented here demonstrate that by careful design of the nanoparticle surface and by choosing sensor surfaces with terminal –CH3 or –COOH groups, improved sensing (micro)systems with very low non-specific adsorption can be obtained.

 Please wait while we load your content...
Please wait while we load your content...