Endosulfan induced the arrest of the cell cycle through inhibiting the signal pathway mediated by PKC-α and damaging the cytoskeleton in spermatogonial cells of mice in vitro
Abstract
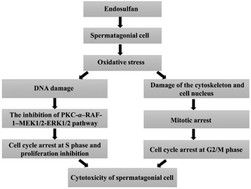
Previous studies have shown that endosulfan has adverse effects on the male reproductive system and the oxidative stress induced by endosulfan is related to its toxicity. However, the molecular mechanism of endosulfan reproductive toxicity is still unknown. To investigate its mechanism of toxicity, GC-1spg cells were exposed to 0, 6, 12 and 24 μg mL−1 endosulfan for 24 h, respectively. The results showed that endosulfan resulted in a dose-dependent reduction in cell viability and increases in the LDH release, apoptosis rate, malondialdehyde (MDA) level, reactive oxygen species (ROS) production and degree of DNA damage including the percentage of tail DNA, tail length, tail moment (TM) and Olive tail moment (OTM). Endosulfan induced the arrest of both S and G2/M phases and proliferation inhibition. The expression of PKC-α, CDK2, cyclin E, RAF-1, MEK1/2, p-MEK1/2, ERK1/2 and p-ERK1/2 in GC-1spg cells declined remarkably after treatment with endosulfan. Endosulfan also damaged the microfilaments, microtubules and cell nuclei, and blocked the mitosis process. The results suggested that endosulfan could induce the cell cycle arrest and proliferation inhibition by inhibiting the protein expression of the cellular signaling pathway mediated by PKC-α because of DNA damage resulting from oxidative stress; meanwhile, endosulfan could also lead to mitotic arrest through direct damage to the cytoskeleton and cell nuclei resulting from oxidative stress, which therefore induced cytotoxicity of GC-1spg cells.

 Please wait while we load your content...
Please wait while we load your content...