Strong CIE activity, multi-stimuli-responsive fluorescence and data storage application of new diphenyl maleimide derivatives†
Abstract
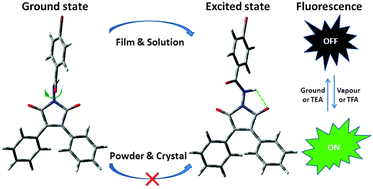
A series of benzamide-based diphenyl maleimide derivatives 2a–d with strong crystallization-induced emission (CIE) activity were synthesized and investigated. 2a–d are almost non-emissive in solutions and amorphous films, but intensely and greenly emissive in crystalline powders and crystals with a quantum yield of 53% and 80%, respectively. The results of X-ray diffraction analysis and theoretical calculation demonstrate that the intramolecular hydrogen bond between C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and NH groups formed in the excited state of 2 quenches the fluorescence, resulting from the rotation of benzamide groups in their solution and amorphous states. However, it would not happen in their crystalline sates for the locked conformation. Thus, the formation and inhibition of the hydrogen bonding in the excited state may be responsible for their CIE activity. Interestingly, the florescence of the 2d solid can be switched between on/off states by external stimuli including grind, heat, solvent vapor, and acid/alkali, which was utilized to develop three kinds of new technologies for rewritable information storage and security ink.
O and NH groups formed in the excited state of 2 quenches the fluorescence, resulting from the rotation of benzamide groups in their solution and amorphous states. However, it would not happen in their crystalline sates for the locked conformation. Thus, the formation and inhibition of the hydrogen bonding in the excited state may be responsible for their CIE activity. Interestingly, the florescence of the 2d solid can be switched between on/off states by external stimuli including grind, heat, solvent vapor, and acid/alkali, which was utilized to develop three kinds of new technologies for rewritable information storage and security ink.

 Please wait while we load your content...
Please wait while we load your content...