Effects of fluorination on the electrochromic performance of benzothiadiazole-based donor–acceptor copolymers†
Abstract
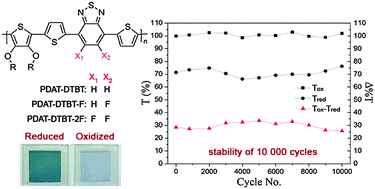
A series of thiophene and benzothiadiazole-based copolymers (PDAT-DTBT) is synthesized through Stille coupling polymerization with two mono- and di-fluorinated benzothiadiazole analogues: PDAT-DTBT-F (1F) and PDAT-DTBT-2F (2F). The introduction of fluorine atoms onto the conjugated polymer backbone is found to have a pronounced effect on the optical, electrochemical and morphological properties, which in turn, influences the electrochromic performance of the fabricated absorption/transmission type devices greatly. All the polymers switch reversibly between the colored neutral states (green/blue) to transmissive oxidized states. Systematic enhancement in the reduction process to sub-second speeds (<0.5 s), high coloration efficiency (748 cm2 C−1) and substantially improved ambient stability are observed upon fluorination of electron acceptors. Long-term stability testing of the PDAT-DTBT-F electrochromic device is carried out for up to 10 000 repeated redox cycles between the applied potentials of +1.6 and −1.6 V, without the observation of significant degradation.

 Please wait while we load your content...
Please wait while we load your content...