The important role of the location of the alkoxy group on the thiophene ring in designing efficient organic nonlinear optical materials based on double-donor chromophores†
Abstract
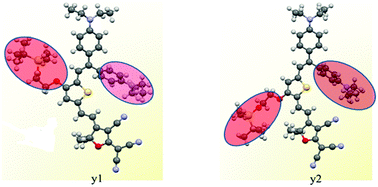
Two novel chromophores y1 and y2 based on the same double donors and the tricyanofuran acceptor linked together via the modified thiophene π-conjugation have been synthesized and systematically investigated. Density functional theory was used to calculate the energy gaps and first-order hyperpolarizability (β) of the two chromophores. Besides, cyclic voltammetry (CV) experiments were performed to determine the different redox properties. The electro-optic coefficient of poled films containing 25 wt% of chromophore y1 and y2 doped in amorphous polycarbonate afforded values of 163 and 105 pm V−1 at 1310 nm, respectively. The obvious different results indicated that the position of the oxygen atom on the thiophene ring is essential to the linear and nonlinear optical properties, with the best results obtained when the alkoxy group was closer to the donor moiety. These results are useful in designing the nonlinear optical materials and will provide new guidance for the future research.

 Please wait while we load your content...
Please wait while we load your content...