Benzobisthiadiazole-based conjugated donor–acceptor polymers for organic thin film transistors: effects of π-conjugated bridges on ambipolar transport†
Abstract
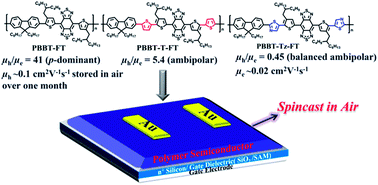
A new series of benzobisthiadiazole (BBT)-based donor–acceptor copolymers, namely, PBBT-FT, PBBT-T-FT, and PBBT-Tz-FT, with different π-conjugated bridges have been developed for polymer thin film transistors (TFTs). It was found that inserting different π-conjugated bridges into the backbone of the polymer allowed tailoring of opto-electrical properties, molecular organizations, and accordingly, ambipolar transport of TFTs. The UV-vis-NIR spectra of all three polymers were similar with the low band gaps of around 1.1 eV. While the lowest unoccupied molecular orbital (LUMO) energy levels were also similar (around −3.8 eV), the highest occupied molecular orbital (HOMO) energy levels varied from −5.05 to −5.42 eV because of the different π-conjugated bridges; moreover, their TFTs exhibited different ambipolar transport. p-Type dominant TFT performances with the hole mobility (μh) reaching 0.13 cm2 V−1 s−1 were observed for the prototype polymer PBBT-FT. However, the device based on PBBT-T-FT with thiophene bridges displayed lower but more balanced hole (μh) and electron (μe) mobilities of 6.5 × 10−3 and 1.2 × 10−3 cm2 V−1 s−1, respectively. The device based on PBBT-Tz-FT with the thiazole units exhibited more evenly balanced hole and electron mobilities (μh/μe = 0.45) along with a significantly enhanced μe ∼0.02 cm2 V−1 s−1. These different semiconducting features were ascribed to different molecular orientations and film morphologies revealed by wide-angle X-ray scattering (WAXS) and atomic force microscopy (AFM).

 Please wait while we load your content...
Please wait while we load your content...