Self-assembly of metal ion induced highly emissive fluorophore-triphenylamine nanostructures: enhanced two-photon action cross-section for bioimaging applications†
Abstract
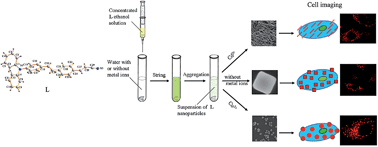
A D–π–A type triphenylamine derivative (E)-3-(4-(4-(diphenylamino)styryl)-phenyl)acrylonitrile (abbreviated as L) is designed, synthesized and characterized by single crystal X-ray diffraction analysis, which is demonstrated to exhibit superior AIEE (aggregation-induced enhanced emission) properties. Self-aggregates of L obtained with and without the guidance of metal ions (Cd2+ or Cu2+) showed morphology-dependent luminescent properties. The coupling and synergy effects between L and metal ions bring about an energy transfer process between the components, which further results in red-shifted absorption and fluorescence, an enhanced fluorescent quantum yield and tunable FL lifetime. The interactions between L and metal ions also change nonlinear optical properties, including two-photon excited fluorescence (2PEF), two-photon absorption (2PA) cross-section (δ), two-photon action cross-section (ηδ) and two-photon absorption coefficient (β), which further leads to successful application of the samples as a two-photon fluorescent probe for labelling the intercellular section. The study indicates that the preparation of nanohybrids from metal ions and an AIEE organic compound is a promising method to prepare 2PA probes possessing a high ηδ for biological imaging.

 Please wait while we load your content...
Please wait while we load your content...