Enhanced DNA release from disulfide-containing layered nanocomplexes by heparin-electrostatic competition†
Abstract
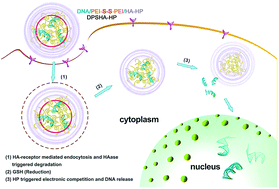
Polycationic vectors are often used to deliver DNA for cancer therapies, but their inefficiency in releasing DNA from the polyplexes after endosomal escape limits DNA transcription and their efficient application in vivo. In this study, DNA/PEI polyplexes were cross-linked by a reduction-sensitive disulfide bond and then further complexed with electrostatic competitive heparin (HP) and hyaluronidase (HAase)-sensitive hyaluronate (HA) to obtain DNA/PEIS/HA–HP (DPSHA–HP). DPSHA–HP was stable in an extracellular environment (pH = 7.4) and degraded by HAase after targeted HA receptor CD44-mediated cell endocytosis, causing the outer shielding of the nanocomplex to loosen. The resulting partially exposed disulfide-linked DNA/PEI nanocomplexes efficiently ruptured the endosome, facilitating the cleavage of disulfide bonds and the release of DNA/PEI polyplexes into the cytoplasm, where DNA release from the polyplexes was remarkably enhanced due to strong electrostatic competition of HP with PEI. Consequently, DPSHA–HP exhibited excellent DNA transfection of the target cells, better than disulfide cross-linked DNA/PEI (25 kDa) and DNA/PEIS/HA. Moreover, these novel layered nanocomplexes have high efficiency in down-regulating B-cell-specific Moloney murine leukemia virus insertion site 1 (Bmi-1) and exhibit significant inhibition of tumor formation with minimal toxicity in a mouse tumor model.

 Please wait while we load your content...
Please wait while we load your content...