Intriguing differences in hydrogen adsorption in CPO-27 materials induced by metal substitution†
Abstract
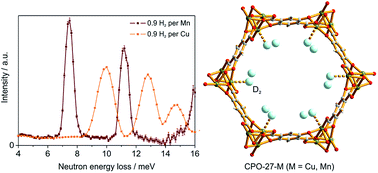
An extraordinarily interesting series of metal–organic framework compounds are the isostructural microporous coordination polymers [M2(dhtp)] (CPO-27–M, M–MOF-74 or M2(dobdc)) in which a high concentration of coordinatively unsaturated metal sites results in high initial heats of adsorption for a variety of adsorbents. We present here a comparative study of hydrogen gas adsorption experiments on CPO-27–Cu and –Mn, which show significant differences in their hydrogen uptake behaviours which can be attributed to the difference in interaction between hydrogen and the respective metal cation incorporated in the framework structure. Inelastic neutron scattering and neutron diffraction experiments were carried out to gain additional insight into the adsorption processes leading to the difference in hydrogen uptake behaviour by the two compounds. On the basis of the experimental results the hydrogen uptake properties of CPO-27–Cu and –Mn are compared, and finally related to the properties of the other members of the CPO-27 series. It is found that CPO-27–Cu demonstrates the lowest isosteric heat of adsorption for H2 of all the CPO-27–M materials reported to date, where M = Ni, Co, Mg, Zn, Mn, and Fe, whereas CPO-27–Mn demonstrates the second lowest. While all the previously reported CPO-27 materials show two steps in the adsorption isotherm and two distinct values corresponding to the first and second adsorption sites in the heats of adsorption, these are not observed for CPO-27–Cu. Consequently, the open metal site and the second adsorption site are energetically equivalent, and there is no preference for the hydrogen gas at the open metal centre.

 Please wait while we load your content...
Please wait while we load your content...