Development of MgAl2O4-stabilized, Cu-doped, Fe2O3-based oxygen carriers for thermochemical water-splitting†
Abstract
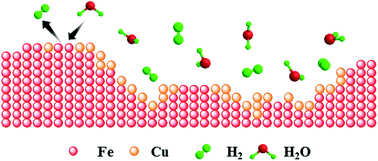
The commercially dominating technology for hydrogen production (i.e. steam methane reforming) emits large quantities of CO2 into the atmosphere. On the other hand, emerging thermochemical water splitting cycles allow the production of a pure stream of H2 while simultaneously capturing CO2. From a thermodynamic point of view, the Fe2O3–Fe couple is arguably the most attractive candidate for thermochemical water splitting owing to its high H2 yield and H2 equilibrium partial pressure. However, the low reactivity of Fe2O3 with methane and the high activity of Fe for methane decomposition (leading to carbon deposition and in turn COx poisoning of the H2 stream) are major drawbacks. Here, we report the development of MgAl2O4-stabilized, Cu-modified, Fe2O3-based redox materials for thermochemical water-splitting that show a high reactivity towards CH4 and low rates of carbon deposition. To elucidate the effect of Cu doping on reducing significantly the rate of carbon deposition (while not affecting negatively the high redox activity of the material) extended X-ray absorption fine structure spectroscopy and energy dispersive X-ray spectroscopy was employed.

 Please wait while we load your content...
Please wait while we load your content...