A conductive selenized polyacrylonitrile cathode material for re-chargeable lithium batteries with long cycle life†
Abstract
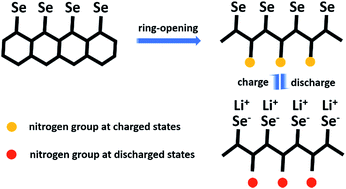
A conductive heterocyclic selenized polyacrylonitrile compound is designed and successfully synthesized by the dehydrogenation/selenation method at high temperature. The selenized polymer materials are examined as cathodes for lithium batteries whose electrochemical behavior resembles but is not totally the same as that of small selenium molecules with no shuttle effect. The periodic variation of the nitrogen response is observed in the XPS spectra of cathode samples with cycling. A new electrochemical reaction mechanism suggesting that nitrogen groups in the selenized polymer structure may also participate in the reversible discharge/charge reactions is proposed and demonstrated for the first time. The selenized polymer cathode delivers a reversible specific capacity of about 350 mA h g−1 after 1000 cycles at a current density of 0.5 mA cm−2 with a total capacity decay of 0.57% (based on the capacity of the 2nd cycle) and a coulombic efficiency of nearly 100%. A specific capacity of 300 mA h g−1 is obtained even when cycled at an ultrahigh current density of 5 mA cm−2, showing the excellent fast discharge/charge capability of the cathodes. Along with a high energy density of 756 W h kg−1, the selenized polyacrylonitrile cathode possesses great application potential in lithium batteries.

 Please wait while we load your content...
Please wait while we load your content...