In situ co-pyrolysis fabrication of CeO2/g-C3N4 n–n type heterojunction for synchronously promoting photo-induced oxidation and reduction properties
Abstract
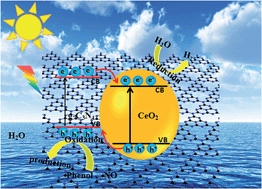
Development of efficient photocatalysts with both photoinduced oxidation and reduction properties is of great importance for environmental and energy applications. Herein, we report the fabrication of CeO2/g-C3N4 hybrid materials by a simple in situ co-pyrolysis method using Ce(IO3)3 and melamine as precursors. The CeO2/g-C3N4 composite catalysts possess outstanding photocatalytic activity for phenol degradation and NO removal under visible light irradiation. The degradation efficiency reaches up to 68.5 and 17.3 times higher than that of pure CeO2 and g-C3N4, respectively. Significantly, it simultaneously exhibits an enhanced hydrogen production rate, which is 1.5 times that of the pure g-C3N4. The highly enhanced photo-induced oxidation and reduction activity could be attributed to the construction of a CeO2/g-C3N4 n–n type heterojunction established by our in situ co-pyrolysis route, which enables intimate interaction across the phase interfaces; this facilitates separation and transfer of photoexcited charge carriers. This study could not only provide a facile and general approach to the fabrication of high-performance carbon-nitride-based photocatalytic materials, but also increase our understanding further on designing new hybrid composite photocatalysts for multi-functional applications.

 Please wait while we load your content...
Please wait while we load your content...