One-step synthesis of cobalt and nitrogen co-doped carbon nanotubes and their catalytic activity for the oxygen reduction reaction†
Abstract
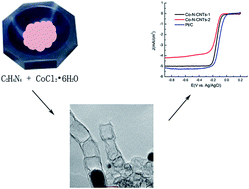
In this paper, cobalt and nitrogen co-doped carbon nanotubes were successfully synthesized by a simple thermal treatment method with dicyandiamide and cobalt(II) chloride hexahydrate. The as-synthesized nanostructures presented excellent catalytic activity and stability toward the oxygen reduction reaction in alkaline solution. Especially, when the molar ratio between dicyandiamide and cobalt(II) chloride hexahydrate was 22 : 1, the catalyst showed a close onset potential, half-wave potentials and limiting current density compared to the commercial platinum/carbon catalyst, which might be attributable to the formation of pyridinic and quaternary nitrogen functional groups as well as the incorporation of cobalt during the thermal treatment. Moreover, the as-synthesized catalyst exhibited good tolerance to the methanol crossover effect.

 Please wait while we load your content...
Please wait while we load your content...