Gold nanoparticle-catalysed photosensitized water reduction for hydrogen generation†
Abstract
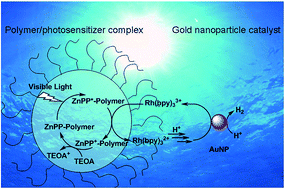
The use of gold nanoparticles (AuNPs) as a catalyst in photosensitized water reduction for hydrogen generation was investigated using conventional three-component systems. Three photosensitizers, ZnPP, ZnTPP and Eosin Y, were utilized to prepare water splitting solutions containing AuNPs (2 nm or 6 nm in diameter), a redox mediator (Rh(bpy)3Cl3), a sacrificial electron donor (TEOA) and an amphiphilic diblock copolymer, PAA52-b-P4VP90, whose micelles can solubilize the hydrophobic ZnPP or ZnTPP. It was found that AuNPs exhibit good catalytic activity comparable to a Pt catalyst (K2PtCl4). Under visible light irradiation at room temperature, hydrogen evolution was observed over the investigated period of time up to 10 hours, with the accumulative amount of the gas being influenced by the size of AuNPs (more efficient with 2 nm AuNPs) and the nature of the photosensitizer (comparable performance with ZnPP and Eosin Y, but lower with ZnTPP). Moreover, absorption and emission spectral changes of ZnPP in the water splitting solution were monitored and analysed; an unidentified species formed at the beginning of irradiation appears to not interfere with the photosensitized hydrogen generation process. This study suggests that AuNPs could be used to replace Pt in catalysing the photosensitized water reduction reaction in widely adopted three-component water splitting systems.

 Please wait while we load your content...
Please wait while we load your content...