Sulfuration–desulfuration reaction sensing effect of intrinsic ZnO nanowires for high-performance H2S detection
Abstract
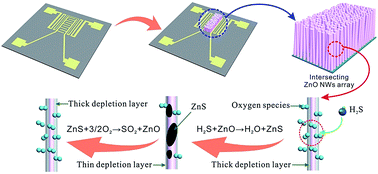
This study explores a novel H2S sensing-effect of an intrinsic (i.e., uncatalyzed) ZnO nanowire (NW) array chemiresistive sensing material. A bottom-up constructed dense array of ZnO NWs with a diameter of 50–60 nm is found and verified to be sulfurized by H2S to form ZnS that can be later desulfurized back to ZnO by ambient oxygen, which is different from the conventional ZnO sensing mechanism where the resistance of semiconductor ZnO is changed via electron depletion layer variation by surface adsorbed oxygen. When H2S is introduced into this ZnO NW sensor, the sulfuration-reaction produced ZnS brings about a dramatic enhancement of sensing performances like sensitivity, limits-of-detection and selectivity. After H2S removal, the desulfuration-reaction helps to realize rapid signal recovery that significantly improves the repeatability of the sensor. Thanks to the nanoscale size effect induced new sensing mechanism, intrinsic ZnO-NWs experimentally exhibit low-temperature (150 °C) high-performance detection toward H2S of ppb to ppm concentration, which could not be realized before even if catalyst-enhanced ZnO was used for H2S sensing at higher temperature.


 Please wait while we load your content...
Please wait while we load your content...