Synthesis of titanium cross-linked chitosan composite for efficient adsorption and detoxification of hexavalent chromium from water
Abstract
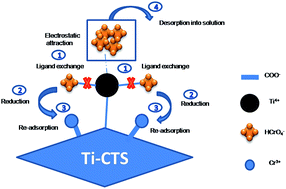
For the simultaneous adsorption and detoxification of hexavalent chromium from water, a new titanium–chitosan (Ti–CTS) composite was synthesized through a metal-binding reaction between titanium ions and the chitosan biopolymer followed by cross-linking with glutaraldehyde. The resultant composite was characterized by FT-IR, XRD, elemental mapping, SEM and XPS. The adsorption properties toward Cr(VI) were systematically investigated as a function of pH, dosage, initial concentration, contact time, temperature and co-existing ions. Experimental data were well described by the Langmuir isotherm and the pseudo-second order model with the maximum adsorption capacity of 171 mg g−1. More attractively, the Cr(VI) could be effectively adsorbed and reduced to the less toxic Cr(III) by the Ti–CTS composite. The experimental results, FT-IR and XPS indicated that the possible removal mechanism of Cr(VI) onto the Ti–CTS composite was summarized into three steps: (i) Cr(VI) adsorption by electrostatic attraction (Ti4+ and HCrO4−) and ligand exchange (Cl− and HCrO4−); (ii) Cr(VI) partly reduced to Cr(III); (iii) the re-adsorption of Cr(III) onto the Ti–CTS composite.

 Please wait while we load your content...
Please wait while we load your content...