Supramolecular hydrogels constructed by red-light-responsive host–guest interactions for photo-controlled protein release in deep tissue†
Abstract
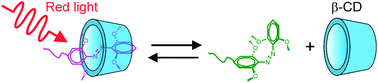
We report a novel red-light-responsive supramolecule. The tetra-ortho-methoxy-substituted azobenzene (mAzo) and β-cyclodextrin (β-CD) spontaneously formed a supramolecular complex. The substituted methoxy groups shifted the responsive wavelength of the azo group to the red light region, which is in the therapeutic window and desirable for biomedical applications. Red light induced the isomerization of mAzo and the disassembly of the mAzo/β-CD supramolecular complex. We synthesized a mAzo-functionalized polymer and a β-CD-functionalized polymer. Mixing the two polymers in an aqueous solution generated a supramolecular hydrogel. Red light irradiation induced a gel-to-sol transition as a result of the disassembly of the mAzo/β-CD complexes. Proteins were loaded in the hydrogel. Red light could control protein release from the hydrogel in tissue due to its deep penetration depth in tissue. We envision the use of red-light-responsive supramolecules for deep-tissue biomedical applications.

 Please wait while we load your content...
Please wait while we load your content...