The divergent effects of strong NHC donation in catalysis†
Abstract
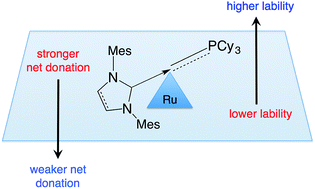
Strong σ-donation from NHC ligands (NHC = N-heterocyclic carbene) is shown to have profoundly conflicting consequences for the reactivity of transition-metal catalysts. Such donation is regarded as central to high catalyst activity in many contexts, of which the second-generation Grubbs metathesis catalysts (RuCl2(NHC)(PCy3)(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHPh), GII) offer an early, prominent example. Less widely recognized is the dramatically inhibiting impact of NHC ligation on initiation of GII, and on re-entry into the catalytic cycle from the resting-state methylidene species RuCl2(NHC)(PCy3)(
CHPh), GII) offer an early, prominent example. Less widely recognized is the dramatically inhibiting impact of NHC ligation on initiation of GII, and on re-entry into the catalytic cycle from the resting-state methylidene species RuCl2(NHC)(PCy3)(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH2), GIIm. Both GII and the methylidene complexes are activated by dissociation of PCy3. The impact of NHC donicity on the rate of PCy3 loss is explored in a comparison of s-GIIm, vs.u-GIIm, in which the NHC ligand is saturated H2IMes or unsaturated IMes, respectively. PCy3 loss is nearly an order of magnitude slower for the IMes derivative (a difference that is replicated, albeit smaller, for the benzylidene precatalysts GII). Proposed as an overlooked contributor to these rate differences is an increase in the Ru–PCy3 bond strength arising from π-back-donation onto the phosphine ligand. Strong σ-donation from the IMes ligand, coupled with the inability of this unsaturated NHC to participate in significant π-backbonding, amplifies Ru → PCy3 π-back-donation. The resulting increase in Ru–P bond strength greatly inhibits entry into the active cycle. For s-GII, in contrast, the greater π-acceptor capacity of the NHC ligand enables competing Ru → H2IMes back-donation (as confirmed by NOE experiments, which reveal restricted rotation about the Ru–NHC bond for H2IMes, but not IMes). Ru → PCy3 back-donation is thus attenuated in the H2IMes complexes, accounting for the greater lability of the PCy3 ligand in s-GIIm and s-GII. Similarly inhibited initiation is predicted for other metal–NHC catalysts in which a π-acceptor ligand L must be dissociated to permit substrate binding. Conversely, enhanced reactivity can be expected where such L ligands are pure σ-donors. These effects are expected to be particularly dramatic where the NHC ligand has minimal π-acceptor capacity (as in the unsaturated Arduengo carbenes), and in geometries that maximize NHC–M–L orbital interactions.
CH2), GIIm. Both GII and the methylidene complexes are activated by dissociation of PCy3. The impact of NHC donicity on the rate of PCy3 loss is explored in a comparison of s-GIIm, vs.u-GIIm, in which the NHC ligand is saturated H2IMes or unsaturated IMes, respectively. PCy3 loss is nearly an order of magnitude slower for the IMes derivative (a difference that is replicated, albeit smaller, for the benzylidene precatalysts GII). Proposed as an overlooked contributor to these rate differences is an increase in the Ru–PCy3 bond strength arising from π-back-donation onto the phosphine ligand. Strong σ-donation from the IMes ligand, coupled with the inability of this unsaturated NHC to participate in significant π-backbonding, amplifies Ru → PCy3 π-back-donation. The resulting increase in Ru–P bond strength greatly inhibits entry into the active cycle. For s-GII, in contrast, the greater π-acceptor capacity of the NHC ligand enables competing Ru → H2IMes back-donation (as confirmed by NOE experiments, which reveal restricted rotation about the Ru–NHC bond for H2IMes, but not IMes). Ru → PCy3 back-donation is thus attenuated in the H2IMes complexes, accounting for the greater lability of the PCy3 ligand in s-GIIm and s-GII. Similarly inhibited initiation is predicted for other metal–NHC catalysts in which a π-acceptor ligand L must be dissociated to permit substrate binding. Conversely, enhanced reactivity can be expected where such L ligands are pure σ-donors. These effects are expected to be particularly dramatic where the NHC ligand has minimal π-acceptor capacity (as in the unsaturated Arduengo carbenes), and in geometries that maximize NHC–M–L orbital interactions.


 Please wait while we load your content...
Please wait while we load your content...